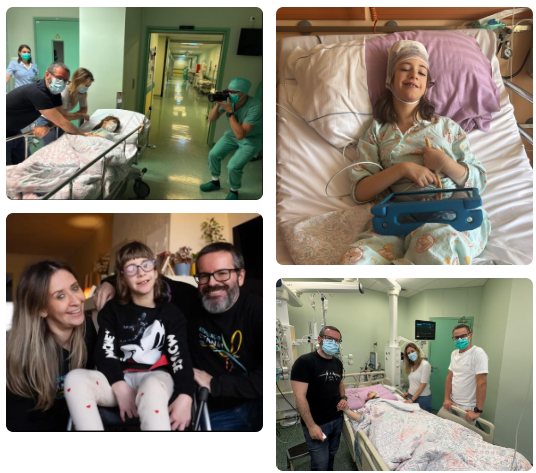
True heroes
continue to smile,
proving that courage
is their superpower.
CTNNB1 Syndrome
CTNNB1 syndrome is a severe neurodevelopmental disorder affecting every aspect of a patient's functioning, including their ability to walk, speak and live independent.
Gene Therapy
URBAGEN is the first CTNNB1 gene therapy showing functional restoration in preclinical models, now advancing toward clinical trials with EMA support.
Dragonfly NHS
Join the global CTNNB1 Natural History Study and help shape future treatments through essential data that supports research, families, and patient care.
Conference 2026
Registration for the 4th International CTNNB1 Syndrome Conference is officially open!
Barcelona, June 18-19 2026
What we do?
The CTNNB1 Foundation is a research-driven non-profit organization focused on developing and supporting gene-related treatments with the potential to benefit patients with CTNNB1 syndrome.
CTNNB1 syndrome will not cure itself. Our team has been working tirelessly to develop a gene replacement therapy and make it available to our young patients as soon as possible.
Newsletter – June 2026
GAIN-CTNNB1 clinical trial open for applications!

We are proud to announce that our clinical trial application has been approved and we have started recruiting participants aged 2-12 years.
For more information, click HERE.
Research Efforts

In 2021, we signed a research agreement with the Children’s Medical Research Institute to start developing different constructs of the gene replacement therapy. Initial preclinical testing was performed with world-renowned laboratories, including The Jackson Laboratory, Charles River, and the National Institute of Chemistry Ljubljana. In November 2023, we initiated the manufacturing process with our amazing partner Viralgen, which produced the clinical grade vector. In 2025 we have completed regulatory enabling toxicology studies with Cima Universidad de Navarra (CIMA) and Czech Centre for Phenogenomics. Biodistribution studies were done at Labena. We are very grateful for all these amazing partnerships.
Our gene replacement therapy product URBAGEN has been granted Orphan Drug Designation (EU/3/25/3101). This recognition by the European Medicines Agency (EMA) is a crucial step on our path to clinical trials and ultimately to an approved treatment for CTNNB1 syndrome.
Just recently, our clinical trial application for the gene therapy has been officially submitted to the EMA. This marks the start of the formal review process and the countdown to patient recruitment has started.
what is gene replacement therapy and
what do we hope to achieve?
Gene replacement therapy is a groundbreaking approach that aims to treat genetic diseases at their root cause. In CTNNB1 syndrome, this therapy delivers a functional CTNNB1 gene directly into the brain, where the majority of symptoms come from.
For children with CTNNB1 syndrome, their existing gene does not produce enough of the β-catenin protein, which is essential for brain development, movement, communication, and learning. Our therapy, URBAGEN, is restoring that critical function.

On our 3rd International CTNNB1 syndrome conference, dr. Damjan Osredkar presented the clinical trial protocol.
Watch it here: https://www.youtube.com/watch?v=PndmKf70rn8
join us in our final
community-driven fundraising campaign
The Final Push for CTNNB1 Gene Therapy
We are calling on every family to reach out to their local community: friends, neighbors, schools, businesses and raise between €1,000 and €5,000.
If just a third of families support this final step, we will reach our goal and move forward without delay.
So far, we have already raised €55,000 thanks to a generous €40,000 donation from the CTNNB1 Association Spain, and an additional €15,000 from two donors (GaletaBio and Sartorius) we personally secured this week to help lead the way in launching this final fundraising campaign.


That leaves us with €445,000 to raise.
You can join this campaign through three main channels:
- Create your own fundraising page on GiveButter and share it with your network: https://givebutter.com/ctnnb1genetherapy
- Companies can donate directly to the CTNNB1 Foundation through a donation agreement (we can help you with this, send email to lavra@ctnnb1-foundation.org).
- CTNNB1 Spain also offers a well-established donation system for Spanish supporters (estimedina@hotmail.com)
- Create your own personalized fundraising brochure to share with your community or workplace in support of our gene therapy program (available in multiple languages!). Send us your child picture and information to lavra@ctnnb1-foundation.org
financial report 2021-2024
Over the years, the CTNNB1 Foundation significantly increased in income, all together more than 4 million euros have been raised or received through many fundraising campaigns and funding opportunities. However, expenses have also risen dramatically, particularly in 2024, due to manufacturing and preclinical testing costs.
Our five largest contributors (2021–2024) have provided 86% of the total funding that is critical to advancing our gene therapy program towards clinical trials.
The “Walking with Urban” campaigns raised €1,462,531 through many Slovenian campaigns, funding mainly basic and proof-of-concept gene therapy studies. The Slovenian government contributed €1,000,000 in late 2024, to enable clinical trial development in Slovenia.
The Slovenian association “Palčica Pomagalčica” raised €580,000, while the founders personally donated €325,000, underlining their strong commitment. CTNNB1 Spain contributed €235,000 through targeted fundraising.
become a CTNNB1 foundation member
As we move closer to the clinical trial – the moment we have all been waiting for – we want to ensure you stay informed and connected.
By becoming a CTNNB1 Foundation Member, you will:
- Receive updates on research and clinical trials
- Stay informed through our monthly newsletter
- Get notified about upcoming events and opportunities to participate
our latest news
Newsletter – June 2026
Newsletter – May 2026
upcoming events
ASGCT Annual Meeting 2026, Boston, USA (May 11–15): https://annualmeeting.asgct.org
- Nina Žakelj – poster presentation
- Damjan Osredkar – poster presentation
- Špela Miroševič – oral presentation
4th International CTNNB1 Syndrome Conference and Clinical Examinations, Barcelona, Spain (June 16–19):
our partners & sponsors